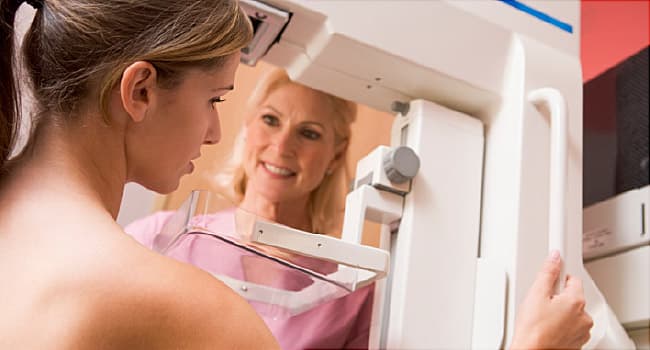
Thermography devices -- also called digital infrared imaging devices -- are approved by the FDA only for use with another screening or diagnostic test like mammography, not as a stand-alone diagnostic tool.
Read More


Thermography devices -- also called digital infrared imaging devices -- are approved by the FDA only for use with another screening or diagnostic test like mammography, not as a stand-alone diagnostic tool.

0 Response to "Don't Be Fooled: Thermography No Substitute for Mammograms, FDA Says"
Post a Comment